|
When electrons want to enter the p-subshell, then the first electron will enter the p x orbital in the clockwise direction. The orbitals are p x, p y, and p z and each orbital can have a maximum of two electrons. Hund’s principle is that electrons in different orbitals with the same energy would be positioned in such a way that they could be in the unpaired state of maximum number and the spin of the unpaired electrons will be one-way.įor example, we already know that the p-subshell has three orbitals. Here we will find out the total number of valence electrons for SF6 by adding the valence electrons for both Sulfur and Fluorine atoms. Hund’s principle is a rule that helps to determine how electrons are distributed in orbitals when multiple orbitals of the same energy level are available. To determine the Lewis Structure of any molecule, we first need to know the total number of valence electrons. That is, the orbital notation of sulfur is 1s 2 2s 2 2p 6 3s 2 3p 4. Now it is possible to find the orbital notation of sulfur very easily through electron configuration. The atomic number of sulfur is 16, which means it has 16 electrons. The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital. This orbital notation system always follows the Aufbau principle. 
This is similar to electron configuration, but numbers are used instead of boxes to represent the positions of the electrons. For the SF6 Lewis structure there are a total of 12 valence electrons on the Sulfur (S) atom. Note that Sulfur (S) is in Period 3 on the periodic table and can have an expanded octet and is able to have more than 8 valence electrons. The sulfur orbital notation is a shorthand system designed to represent the exact positions of the electrons in the sulfur atom. There are a total of 48 valence electrons in the Lewis structure for SF6. 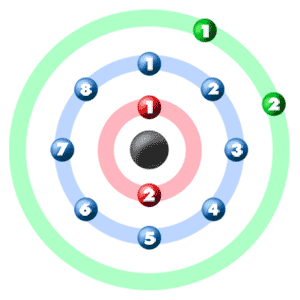
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons. Each orbital can have a maximum of two electrons. The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. The sub-energy levels are known as s, p, d, and f. The sub-energy levels depend on the azimuthal quantum number. The most probable region of electron rotation around the nucleus is called the orbital. These sub-energy levels are also called orbital. Again, atomic energy shells are subdivided into sub-energy levels. These circular paths are called orbit (shell). The electrons of the atom revolve around the nucleus in a certain circular path. To create an orbital diagram of sulfur, you first need to know the atomic orbitals and the orbital notation for the sulfur atom, and also you need to know Hund’s principle. Orbital is the region of space around the nucleus of an atom where electrons are found. This diagram shows how the electrons in the sulfur atom are arranged in different orbitals. The sulfur orbital diagram is a graphical representation of the electron configuration of the sulfur atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
